Firefly and PC GAMESS-related discussion club

Learn how to ask questions correctly

benjoe rey
benz_601@yahoo.com
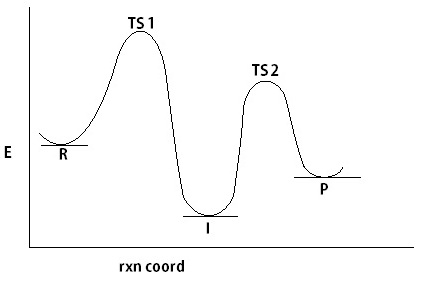
Thanks for the link. But I think that was a different problem. I think the problem there is that a negative Energy of activation was obtained. In my case, I am dealing with a multistep mechanism. If I split these steps into single step mechanisms, each step looks good (i.e., ts has the highest energy, positive Ea, reasonable dE). But in the multistep E profile, it turns out that an intermediate, relative to the reactant and the final products, has a lower energy. (I've attached a rough illustration of the E profile)
Sorry for the trouble.
Regards,
Benjoe
On Sat Oct 9 '10 1:16am, sanya wrote
------------------------------------
>Hi,
>Here you can find a discussion about very similar problem. This may be a hint for you.
>On Fri Oct 8 '10 3:09pm, benjoe rey wrote
>-----------------------------------------
>>Hi everyone,
>>It's me again.
>>Is it odd if in a reaction, the energy of an intermediate is lower than either the reactants and the products? Is there a possibility of that happening, say for a catalyzed reaction? Or is it because a wrong turn along the PES occured? I am so sorry to bother you all.
>>Regards,
>>Benjoe
Energy Diagram